Extend Fertility research concludes GnRH-agonist trigger is an effective option for normal- and low-responding egg freezing patients
One of our goals is not just to treat our own patients, but to use our expertise to improve the fertility field as a whole by providing evidence for best practices in egg freezing, IVF, and beyond. That’s why, in 2018, we founded the Center for Fertility Research and Education, with a mission to close the knowledge gap about fertility and fertility preservation.
Some of our latest research: a study determining the effectiveness of different types of “trigger” medications for normal and low responders (women who produce normal or low numbers of eggs in response to fertility medications). This is the first study to examine how GnRH-agonist triggers work for women doing egg freezing.
The paper, titled “The Use of GnRH-agonist Trigger for the Final Maturation of Oocytes in Normal and Low Responders Undergoing Planned Oocyte Cryopreservation,” was published in Human Reproduction, one of the foremost medical journals for the fertility field, in May.
Background information
We know that women who are high responders—meaning they produce more eggs than normal during an ovarian stimulation cycle—are at higher risk for ovarian hyperstimulation syndrome, or OHSS (though the overall risk is still quite low). OHSS is a condition in which the ovaries respond too excessively to medication, and can become enlarged and swollen with fluid.
We also know, based on previous research, that using a GnRH-agonist trigger such as Lupron can prevent OHSS in high responders. However, limited data exists to support the use of GnRH-agonist triggers in normal or low responders. Additionally, all previous studies were performed on women undergoing in vitro fertilization for infertility—there have been no studies, until now, on the use of GnRH-agonist triggers in non-infertile women doing egg freezing to preserve fertility.
What is a “trigger” medication?
In IVF or egg freezing, a trigger medication is the final shot, taken 36 hours prior to the egg retrieval. It’s referred to as a “trigger” because it prompts the eggs to undergo their final stage of maturation, just in time to be retrieved.
Maturation refers to the final steps of division and development that an egg undergoes before being ovulated. This is an extremely important step in the process, as only mature eggs can be fertilized to create embryos (either now or, if frozen, in the future).
What kind of trigger medication was used in the past?
Historically, fertility physicians used an hCG (human chorionic gonadotropin) trigger medication, such as Ovidrel. During a natural menstrual cycle, you’ll have a spike of luteinizing hormone (LH) just before ovulation; the role of LH is to prompt the egg to mature. Because hCG and LH are chemically similar, your body responds to hCG as it would to luteinizing hormone. So, when you inject hCG at the end of an egg freezing or IVF cycle, it stimulates the eggs to mature and be ovulated within 36 hours.
The problem with hCG is that it lingers in the body for a long time after the egg retrieval, promoting further production of hormones in the ovaries. For those hoping to get pregnant right away through a fresh transfer after IVF, these hormones can support implantation (known as “luteal phase support”).
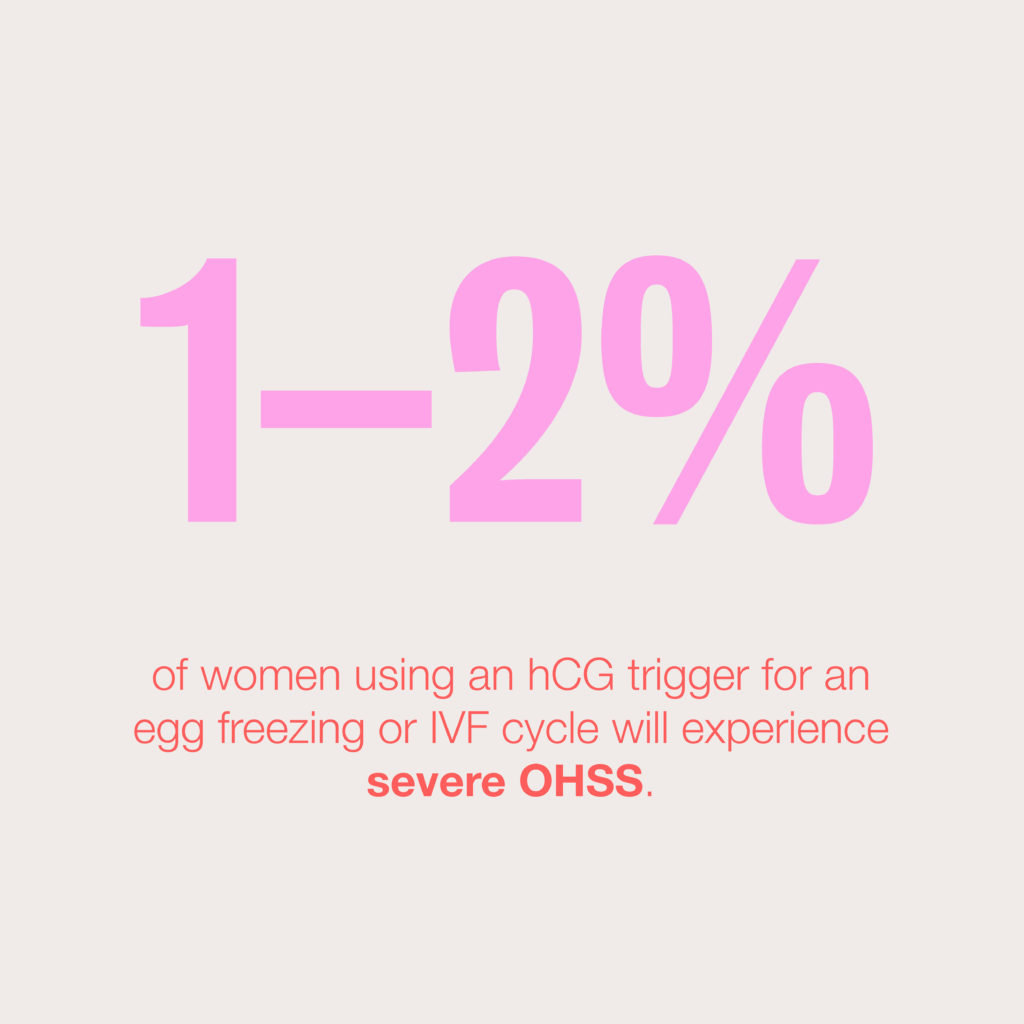
But the extended impact of hCG also increases and prolongs the side effects of ovarian stimulation and increases the risk of OHSS. Research suggests that the chance of severe OHSS following an hCG trigger is approximately 1–2% for IVF patients under age 36.
How is a GnRH-agonist trigger different?
A GnRH agonist is a medication that activates (or “agonizes”) the gonadotropin-releasing hormone receptor in the pituitary gland. This stimulates the pituitary gland to empty its stores of LH and FSH (follicle-stimulating hormone), mimicking the LH/FSH surge that your body naturally experiences during a menstrual cycle. Again, the ovarian follicles’ response to the LH is to initiate maturity and ovulation.
The effect of LH is brief, and the ovaries return to normal quickly after the retrieval. So, while GnRH-agonist triggers, such as leuprolide (brand name Lupron), don’t offer the luteal phase support that hCG offers, they are less likely to cause OHSS. In one study of IVF patients at high risk for OHSS, the use of GnRH-agonist as the primary trigger medication reduced the incidence of OHSS to zero.
This is especially important for women who are freezing their eggs, since they are not trying to get pregnant right away, so the benefits of hCG are not a trade-off for them.

Estradiol or “E2” levels as a measure of ovarian response
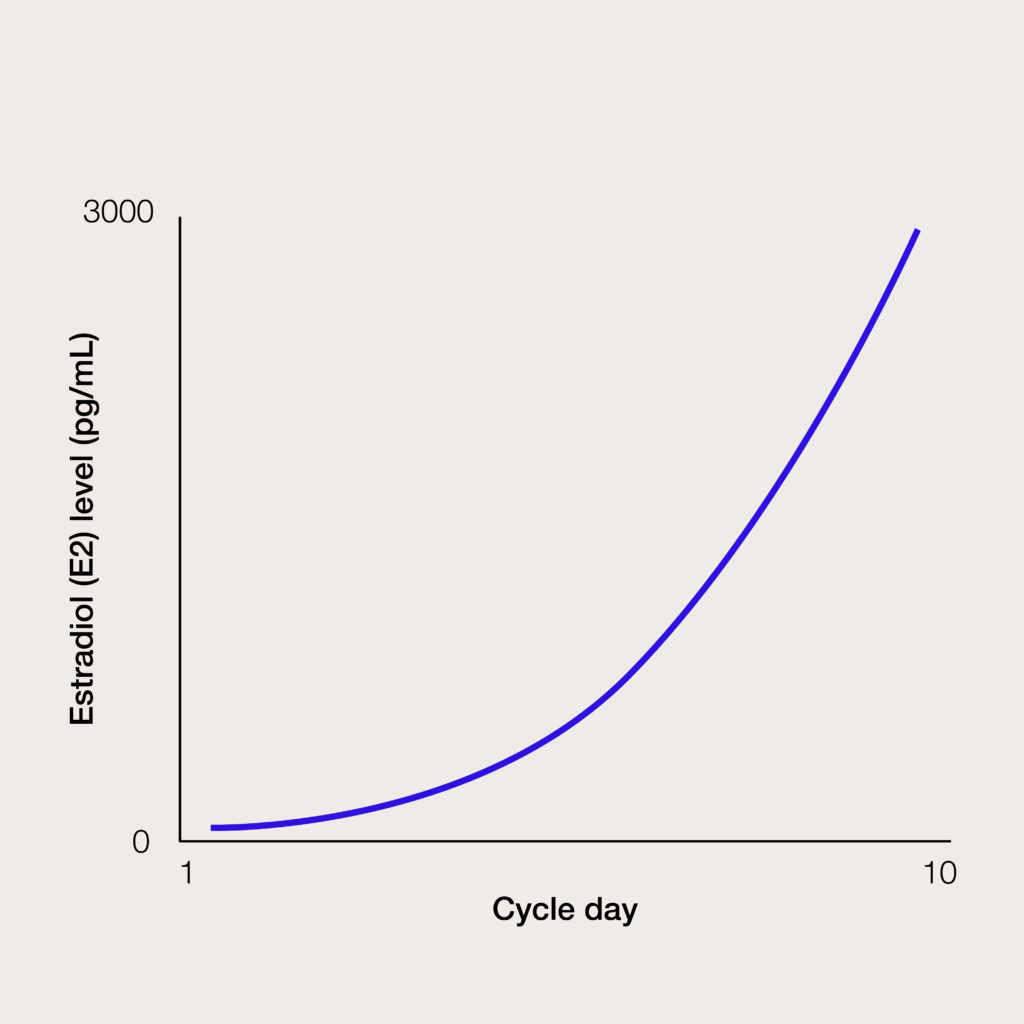
At the beginning of an IVF or egg freezing cycle, levels of estradiol (an estrogen hormone) are quite low; these levels rise rapidly in response to ovarian stimulation medication, as multiple ovarian follicles grow. The increase in E2 levels is an indirect measure of how responsive the ovaries are to the medication.
Patients with peak E2 levels above 3,000 pg/mL are considered high responders, while patients with peak E2 levels below 1,000 pg/mL are considered low responders. High responders are at a higher risk of OHSS, so the majority of research around GnRH-agonist triggers has examined their effectiveness and safety among this population.
What we analyzed: whether a GnRH-agonist trigger is an effective option for normal/low responder egg freezing patients
Our study was a retrospective analysis of 1,680 egg freezing cycles among 1,189 patients. Just over half of the cycles (57.1%) used a GnRH-agonist trigger. We compared the rates of mature eggs across trigger types, as well as between high responders and lower responders.
Results
On average, about 71% of eggs retrieved were found to be mature; among those using GnRH-agonist triggers, the maturity rate was about 73%. This confirms that, for egg freezing patients, a GnRH-agonist reduces the chance of OHSS without reducing the percentage of mature eggs retrieved per cycle.
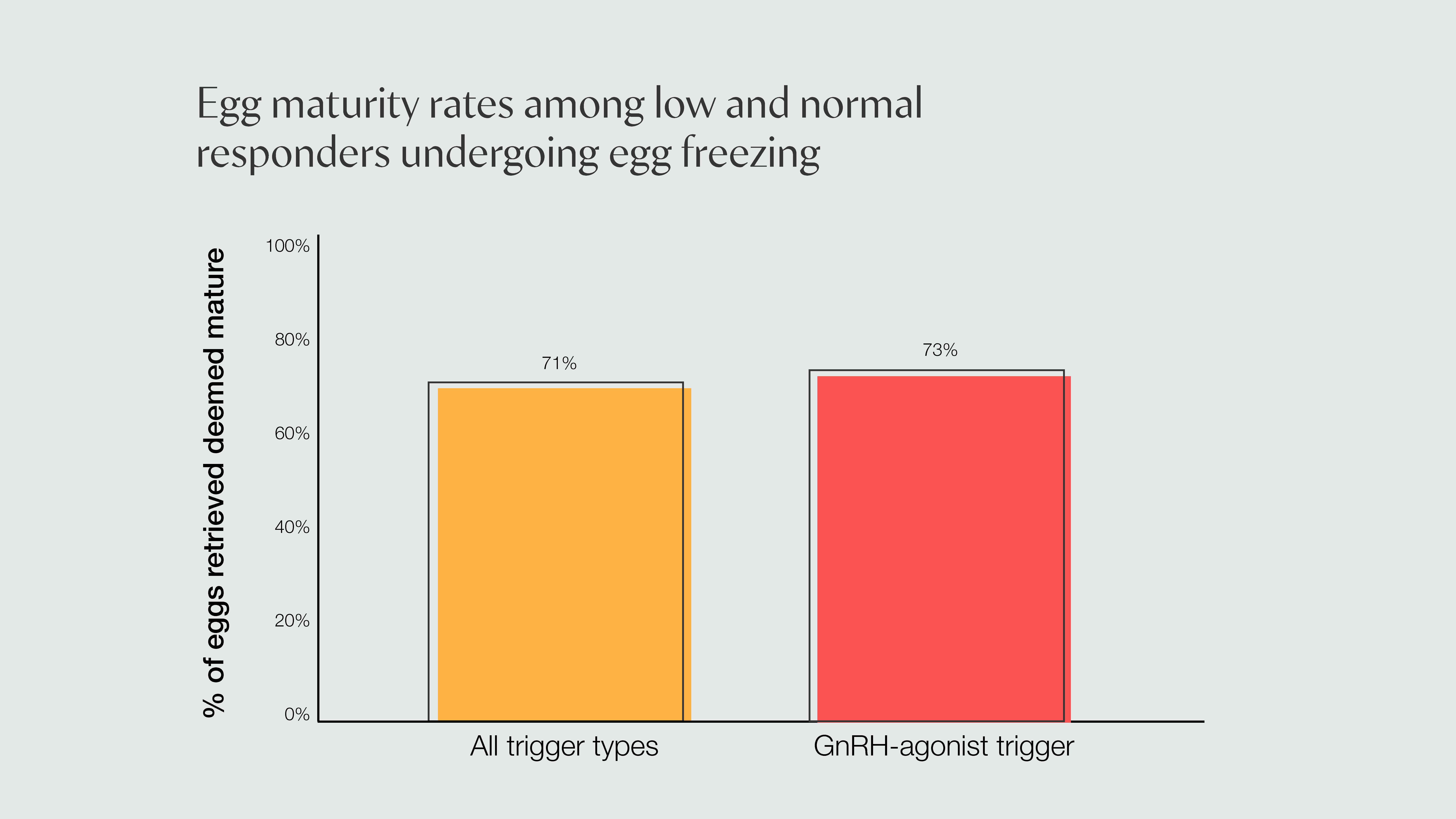
Because GnRH-agonist triggers are We also compared the use of GnRH-agonist triggers in women considered high responders—those with peak E2 levels of 3,000 pg/mL or more—with those whose peak E2 levels were below 3,000 pg/mL. We found that maturity rates were equivalent between the two groups, confirming that GnRH-agonist triggers are equally effective for women not considered high responders as they are for high responders. Low responders, those with peak E2 levels below 1,000 pg/mL, had lower maturity rates, regardless of trigger type.
What else do we need to know?
Because our study specifically analyzed egg freezing cycles, we don’t have data on pregnancy rates from these subjects. Other studies suggest that embryo euploidy rates (the percentage of healthy embryos) are essentially equivalent between GnRH-agonist cycles and hCG cycles, but we’d need a follow-up of egg freezing patients to confirm.
How will this study be used to improve care?
In fertility medicine, and especially in egg freezing, success rates depend on two things: the age of the patient when they retrieve the eggs, and how many eggs they retrieve. That means we’re continuously balancing the need to maximize the result of a cycle—in terms of the number of mature eggs retrieved—with the need to reduce and prevent unpleasant or harmful side effects.
This study has added to the evidence in support of GnRH-agonist triggers as a way to do just that: reduce patients’ chances of OHSS, without reducing the success of their egg freezing cycle. It also highlights that best practices for egg freezing patients may differ from best practices for IVF patients. For too long, research around fertility treatment protocols has focused on IVF patients, and egg freezing patients have simply been lumped together with those trying to conceive—even though their goals and preferences may be different.
As the largest egg freezing program in the country, we’ve implemented this data into our own protocols, and hope this publication will empower other fertility centers to provide the best possible care to their egg freezing patients.